Chapter 4
Д£ (c,+c2) = ζ(c2)-ζ(cl)
∆c 2 c2- c1
[4.18]
Here ζis zeta potential and c is concentration of the additive.
0-----------------ι-----------------ι------. .--------ι----------------4.-.-.-.-.___—
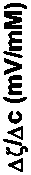
Э-
------
-20
-4° ∣∣
// O Adding NaOH, with Ca/Mg
Adding NaOH, with Ca/Mg
Adding NaOH, without Ca/Mg
Adding carbonate, with Ca/Mg
Adding carbonate, without Ca/Mg
Adding metasilicate, with Ca/Mg _
Adding metasilicate, without Ca/Mg
Adding orthosilicate, with Ca/Mg
Adding orthosilicate, without Ca/Mg -
Adding citrate, with Ca/Mg
Adding citrate, without Ca/Mg
ill O Adding NaOH, without Ca/Mg
'θθ ill ∙ Adding carbonate, with Ca/Mg
∙∣∣ ∙ Adding carbonate, without Ca/Mg
.θQ _ Ш + Adding metasilicate, with Ca/Mg _
!f + Adding metasilicate, without Ca/Mg
is ∙ Adding orthosilicate, with Ca/Mg
-100 j/ * AddingorthosiIicatelWithoutCaZMg-
7 * Adding citrate, with Ca/Mg
♦ Adding citrate, without Ca/Mg
-120----------------------l----------------------l----------------------l----------------------'----------------------
0 0.2 0.4 0.6 0.8 1
Concentration (mM)
Figure 4.17 Zeta potentials change of kaolinite samples in synthetic brine
For hydroxide, citrate and carbonate, zeta potential change per unit additive
concentration is around 15-25 mV∕mM. But for meta- silicate or ortho-silicate,
zeta potential change is much larger than other additives at low concentration
(<10^4 M). And zeta potential change decreases very fast with the increase of
silicate concentration. Compare with other anions, silicate ions have the greatest
effect per unit addition on changing zeta potential of kaolinite.
122
More intriguing information
1. El Mercosur y la integración económica global2. The name is absent
3. Micro-strategies of Contextualization Cross-national Transfer of Socially Responsible Investment
4. Problems of operationalizing the concept of a cost-of-living index
5. Literary criticism as such can perhaps be called the art of rereading.
6. Reconsidering the value of pupil attitudes to studying post-16: a caution for Paul Croll
7. Moffett and rhetoric
8. Olive Tree Farming in Jaen: Situation With the New Cap and Comparison With the Province Income Per Capita.
9. Importing Feminist Criticism
10. Parallel and overlapping Human Immunodeficiency Virus, Hepatitis B and C virus Infections among pregnant women in the Federal Capital Territory, Abuja, Nigeria